The clinical research landscape is evolving faster than ever. Gone are the days when monitoring a clinical trial meant a Clinical Research Associate (CRA) living out of a suitcase and visiting hospital sites five days a week.
Welcome to the era of Risk-Based Quality Management (RBQM)—and with it, the rise of the Centralized Monitoring Assistant (CMA).
If you’re based in Noida, India, or aiming to enter one of the world’s fastest-growing clinical operations hubs, understanding this role could be your gateway to a future-proof clinical research career. Let’s break down why the CMA role has become a cornerstone of modern drug development 🧪
What Exactly Is Centralized Monitoring? 🔍
Centralized monitoring refers to the evaluation of clinical trial data from a central location rather than onsite at the investigator hospital.
Using advanced clinical systems, dashboards, and analytics, centralized teams can:
- Identify trends across hundreds of sites
- Detect outliers or inconsistent data
- Spot missing or delayed entries
- Flag potential patient safety or compliance risks—in real time
This approach doesn’t just improve efficiency—it protects data integrity, regulatory compliance, and patient safety.
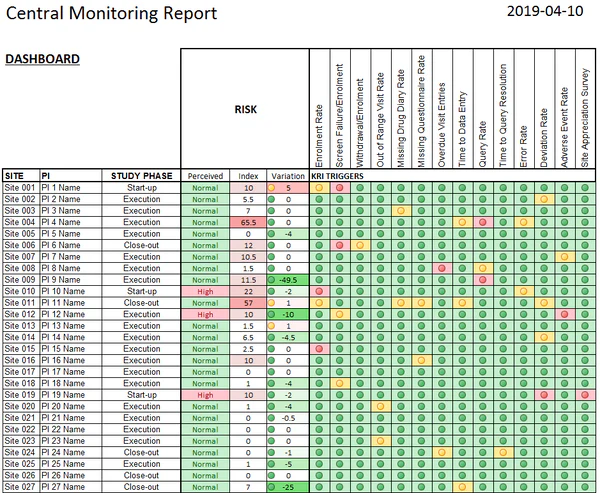
Key Responsibilities: A Day in the Life 🗓️
A Centralized Monitoring Assistant is far more than an administrative role. You’re the “eyes in the sky” for the entire study team.
🔹 System Reporting & Trackers
You’ll run system-generated reports and maintain study trackers that allow Clinical Leads and Project Managers to make informed, strategic decisions 📊
🔹 KRI (Key Risk Indicator) Review
One of the most impactful parts of the role. You’ll review site-level KRIs to identify:
- Underperforming sites
- Data quality inconsistencies
- Potential protocol deviations
🔹 Risk Identification & Escalation
You act as an early warning system, helping teams intervene before minor issues escalate into audit findings or regulatory concerns ⚠️
🔹 Administrative & Study Support
From managing system access requests to preparing i-site packs (site-specific data review packages), you keep the study engine running smoothly.
🔹 Technical Collaboration
You’ll often work closely with Technical Solution Specialists (TSS). If Excel formulas, data visualization, or dashboards excite you—you’ll thrive here 💻
Why Noida Is the Place to Be 🇮🇳
Noida has rapidly emerged as a global hub for Clinical Research Organizations (CROs) and Global Capability Centers (GCCs).
For CMAs, this means:
- Hybrid work models across leading CROs
- Global trial exposure with US, EU, and APAC stakeholders 🌍
- Clear career progression: Centralized Monitoring Assistant → Central Monitor → Clinical Lead → Project / Program Manager
Requirements: Do You Have the “Right Stuff”? 🎓
Recruiters look beyond degrees—they look for the right mindset.
✔ Educational Background
Graduate or Postgraduate degree in:
- Life Sciences
- Pharmacy
- Biotechnology
- Biology or related fields
✔ Experience
- ~2 years of overall work experience
- At least 6 months in clinical research (operations, data review, site support, etc.)
✔ Regulatory Knowledge
Working knowledge of ICH-GCP—the backbone of ethical and compliant clinical trials.
✔ Soft Skills
- Exceptional attention to detail
- Strong written and spoken English
- Professional communication with investigators and site teams 📞
The Secret Sauce: Mastering the Tech 🛠️
To truly stand out, move beyond “MS Office proficiency.”
Centralized monitoring thrives on data interpretation, not just data entry.
Key tools and skills include:
- Predictive analytics (anticipating site or study risks)
- Data integrity & quality review
- Clinical systems such as:
- Medidata Rave
- Veeva Vault
- Oracle InForm
Apply Now 👇🏻
📌 Centralized Monitoring Assistant role at IQVIA
👉 https://jobs.iqvia.com/en/jobs/R1422531-0
Final Thoughts: Is This Career Path for You? 🌟
The Centralized Monitoring Assistant role is ideal if you:
- Love clinical science
- Enjoy analytics and problem-solving
- Prefer an office-based or hybrid role over constant travel
If spotting a strange data trend makes you ask “Why is this happening?”—and you enjoy finding the answer—this role is made for you.
With India’s clinical trial ecosystem expanding rapidly, there’s never been a better time to enter centralized monitoring. It’s high-impact, tech-driven, and places you at the heart of modern medicine 💊✨


