The global healthcare and life sciences industry is rapidly evolving, creating exciting career opportunities for professionals with a scientific background. One such opportunity is the Clinical Data Services Associate (eTMF Management) role at Accenture, a globally recognized leader in consulting, technology, and healthcare solutions.
If you are a Life Sciences graduate with 1–3 years of experience and want to build a career in clinical research, data management, and regulatory documentation, this role could be your perfect next step.
🏢 About Accenture


Accenture is a global professional services company with over 784,000 employees across 120+ countries. It specializes in:
- Strategy & Consulting
- Technology & Digital Transformation
- Operations & Healthcare Solutions
Within the Life Sciences R&D domain, Accenture supports:
- 🔬 Clinical Trials
- 📊 Clinical Data Management
- 💊 Pharmacovigilance
- 📋 Regulatory Affairs
- 🧬 Patient Services
The company plays a crucial role in helping pharmaceutical companies bring treatments to market faster.
📍 Job Details
- Position: Clinical Data Services Associate
- Skill Area: Clinical eTMF Management
- Location: Hyderabad
- Experience: 1–3 Years
- Qualification: Life Sciences Graduate
- Job Type: Full-Time
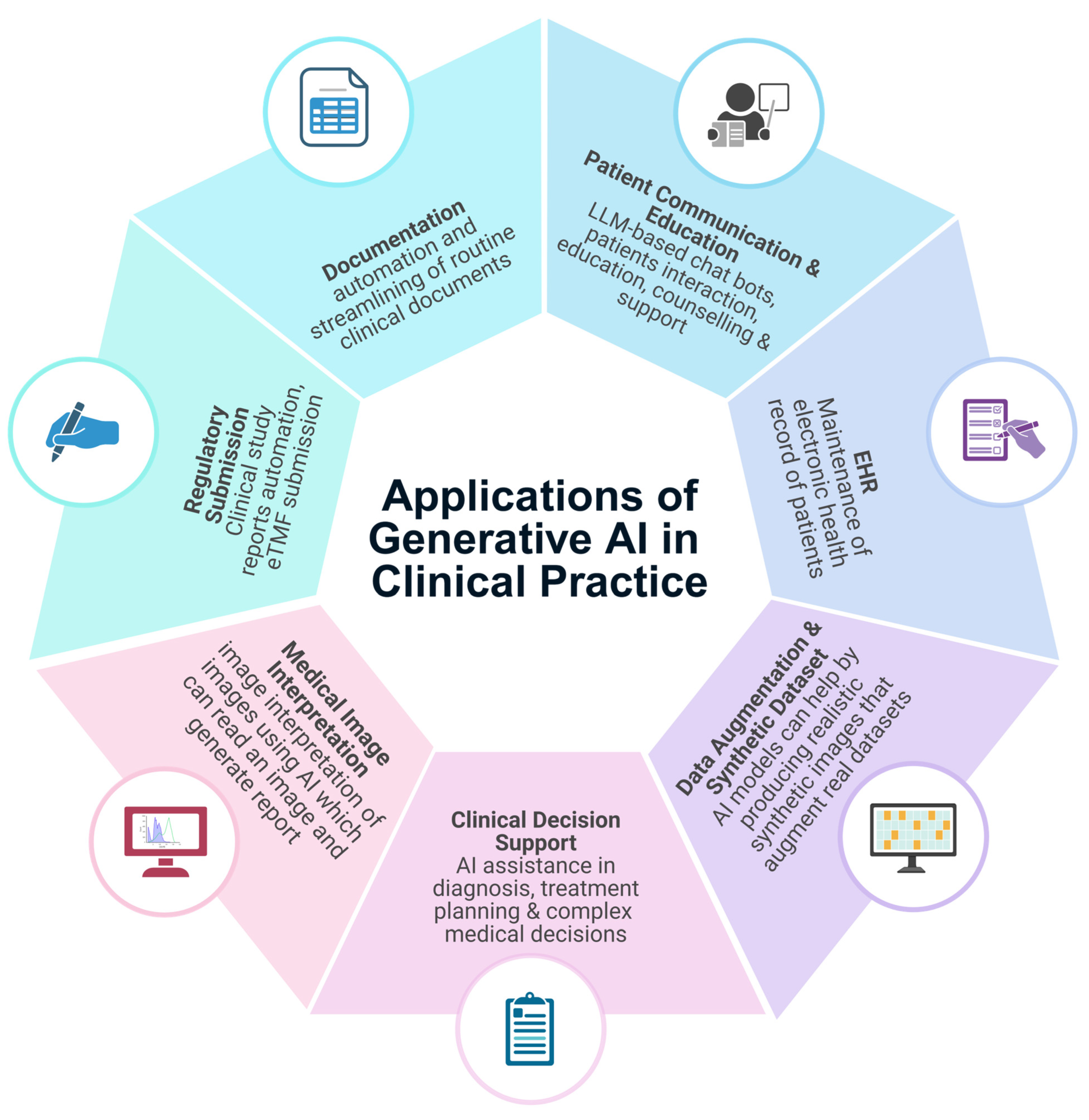
🧾 What is Clinical eTMF?

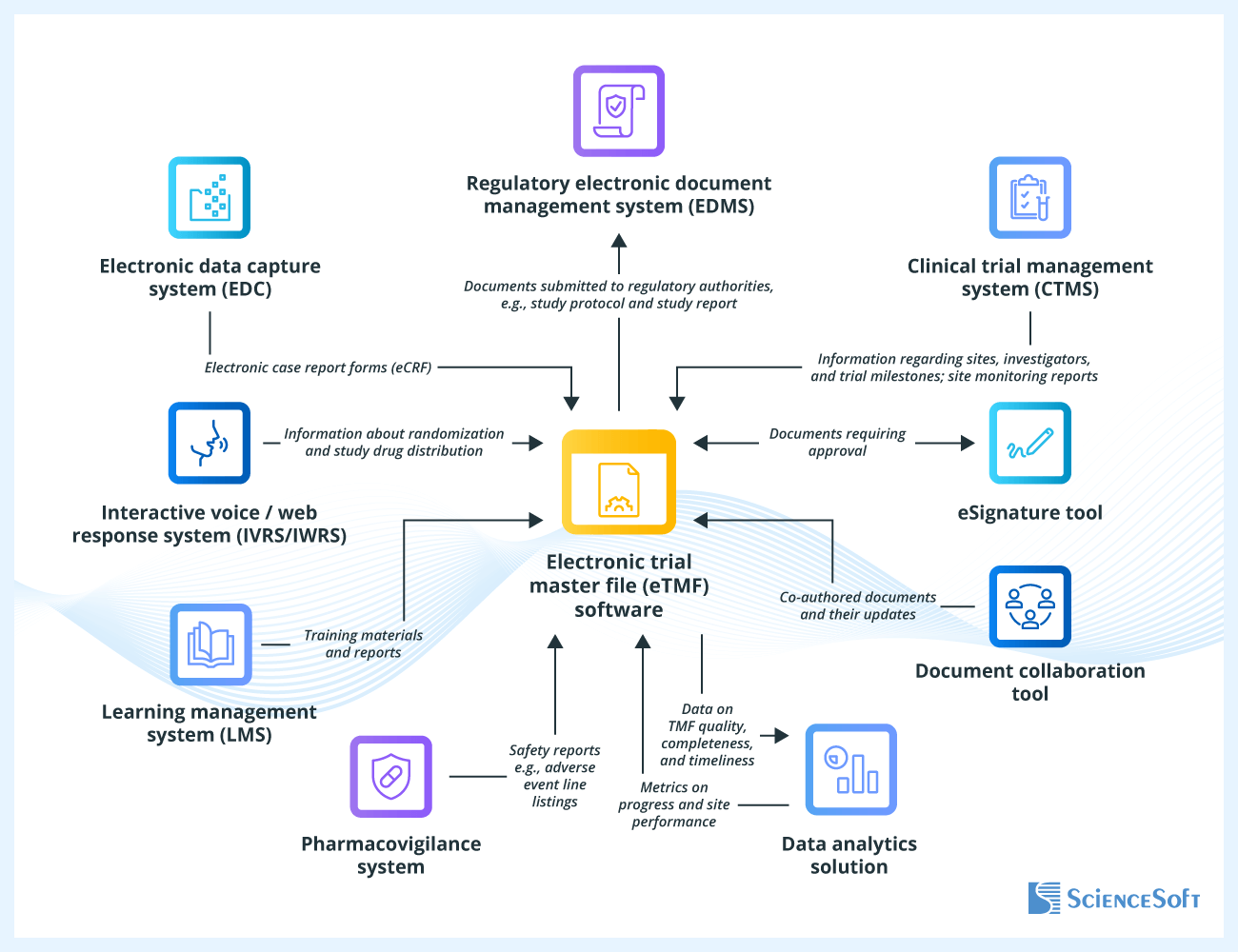
The Electronic Trial Master File (eTMF) is a digital system used to manage essential clinical trial documents.
It ensures that trials are conducted in compliance with:
- Good Clinical Practice (GCP)
- Regulatory guidelines
- Company SOPs
Key Activities:
- 📂 Document organization
- 🔍 Quality review
- 📑 Compliance checks
- ✔️ Audit readiness
- 📊 Structured documentation
💼 Key Responsibilities
📁 TMF Review & Quality Control
- Perform TMF reviews
- Ensure regulatory compliance
📋 Documentation Management
- Protocol documents
- Trial Master File structure
- End-of-trial documentation
- Document reconciliation
🔍 Data Accuracy Checks
Ensure documents are:
- Accurate
- Complete
- Properly indexed
- Audit-ready
🤝 Collaboration
Work with:
- TMF Managers
- Clinical Teams
- Regulatory Teams
- Sponsors
🎯 Skills Required
- ✔ Strong attention to detail
- ✔ Stakeholder management
- ✔ Deadline management
- ✔ Communication skills
- ✔ Problem-solving ability
- ✔ Ability to work under pressure
🎓 Educational Qualification
Eligible candidates include graduates in:
- B.Pharm / M.Pharm
- B.Sc Life Sciences
- Biotechnology
- Microbiology
- Biochemistry
- Zoology / Botany
🚀 Career Growth Opportunities
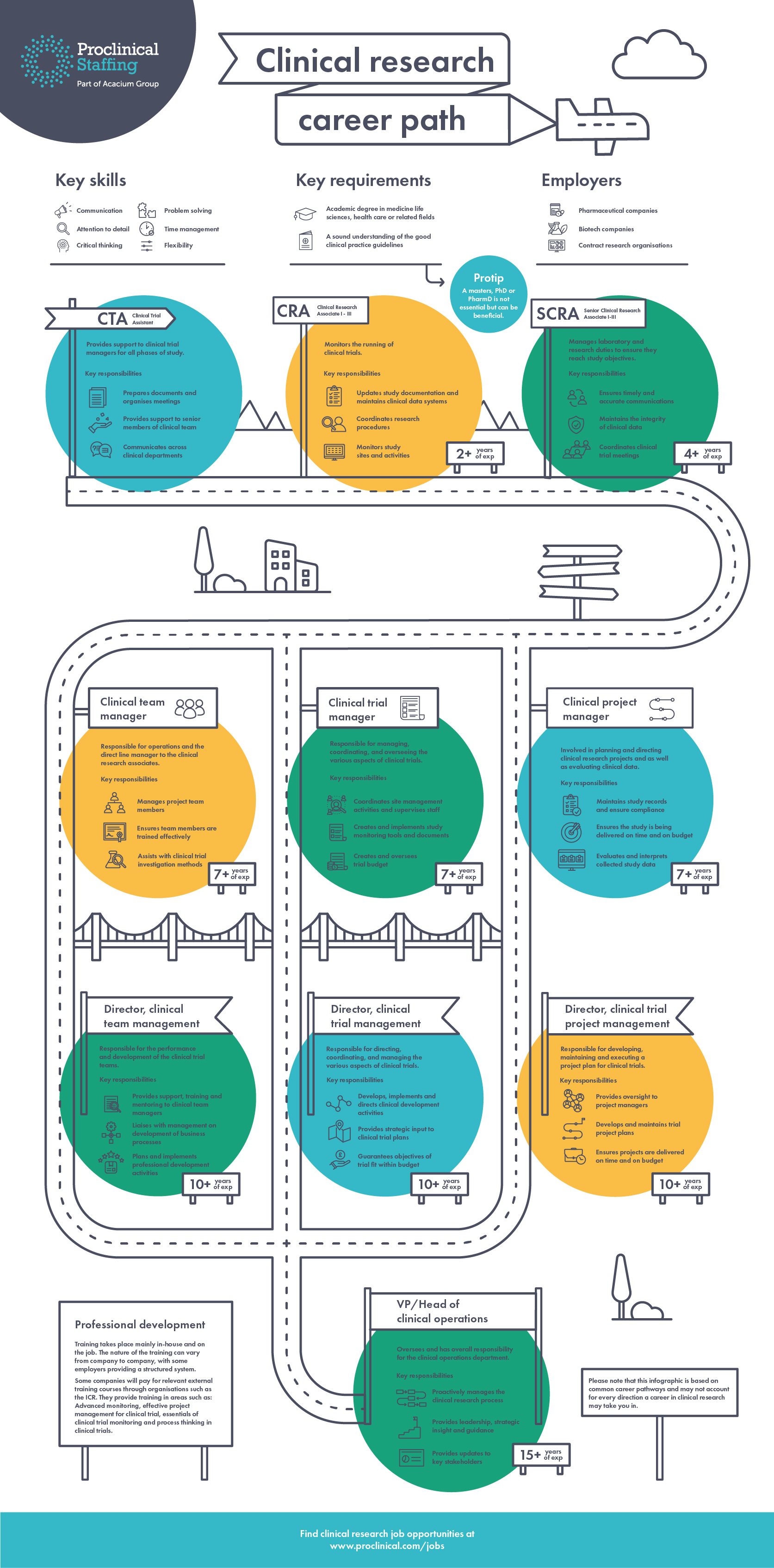


This role opens doors to:
- 📈 Clinical Data Analyst
- 📈 TMF Specialist
- 📈 Clinical Data Manager
- 📈 Clinical Trial Manager
- 📈 Regulatory Affairs Specialist
🌟 Why Work at Accenture?
- 🌍 Global exposure
- 📚 Continuous learning programs
- 🧠 Technology-driven work environment
- 💼 Long-term career stability
📌 Final Thoughts
The Clinical Data Services Associate (eTMF) role at Accenture is an excellent opportunity for life sciences professionals looking to enter or grow in the clinical research and data management field.
With increasing demand for clinical documentation and regulatory compliance experts, this role provides a strong foundation for long-term career growth.