If you are looking to build a career in the clinical research industry, here is an exciting opportunity in Pune. Global language and technology company TransPerfect is currently hiring for the position of Clinical Document Specialist I at its Pune office.
This role is ideal for professionals with clinical research experience who want to work closely with clinical documentation, regulatory processes, and electronic Trial Master File (eTMF) systems.
The position offers valuable exposure to clinical trial documentation management, regulatory compliance, and international clinical study operations.
Below is a complete overview of the job role, responsibilities, eligibility criteria, and application process.
Job Overview
Job Title: Clinical Document Specialist I
Company: TransPerfect
Location: Aundh, Pune, Maharashtra
Job Type: Full-Time (On-site)
Shift Timing: 9:30 AM – 6:30 PM
Work Mode: Work from Office
This role falls under the Localization and Clinical Documentation division, where professionals manage and review clinical trial documents to ensure compliance with regulatory standards and sponsor expectations.
About the Role
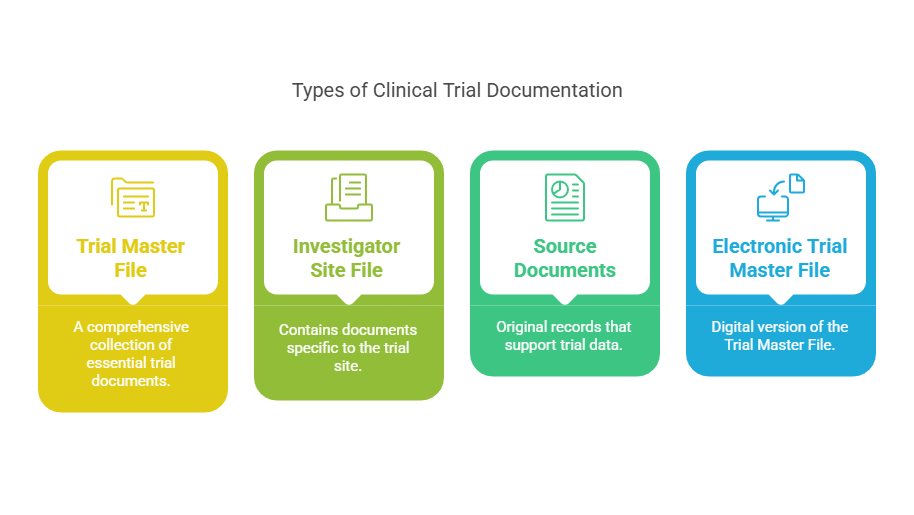
A Clinical Document Specialist plays an important role in maintaining the Trial Master File (TMF) for clinical studies.
The TMF contains essential documents that demonstrate that a clinical trial has been conducted according to regulatory guidelines and Good Clinical Practice (GCP) standards.
In this role, you will:
- Review and manage documents within the electronic Trial Master File (eTMF) system
- Ensure proper indexing and organization of study documentation
- Maintain compliance with regulatory requirements and company SOPs
You will also collaborate with clinical teams, project managers, and document management specialists to ensure that documentation is accurate and audit-ready.
Key Responsibilities
Document Review and Indexing
Review essential and non-essential documents submitted through the eTMF workflow system and ensure they are correctly indexed in the Trial Master File.
Maintain Quality Standards
Ensure all clinical documents meet company SOPs, quality control standards, and productivity targets.
Track Project Documentation
Maintain study-specific trackers to monitor document progress and ensure required files are available for audits or regulatory inspections.
Collaboration with Internal Teams
Work closely with clinical documentation team leads and internal stakeholders to resolve documentation issues.
Support User Acceptance Testing (UAT)
Assist with User Acceptance Testing (UAT) during system updates or new software releases.
Clinical Document Review
Review additional clinical trial documents as assigned by the project team.
Eligibility Criteria
Education
Bachelor’s Degree in:
- Life Sciences
- Pharmacy
- Biotechnology
- Or a related field
Experience
Minimum 2 years of experience in clinical research.
Relevant experience may include:
- Site Management
- Site Start-Up Activities
- eISF (Electronic Investigator Site File)
- Pharmacovigilance
- Clinical Trial Management Systems (CTMS)
- Medical Writing
- Regulatory Submissions
- Clinical Data Management
- HIPAA Compliance
Required Skills
Candidates applying for this role should have:
- Knowledge of clinical development phases and clinical trial processes
- Understanding of ICH-GCP guidelines
- Familiarity with clinical study documentation and essential trial documents
- Strong written and verbal English communication skills
- Ability to work with Microsoft Office tools (Word, Excel, Outlook)
- Interest in learning eClinical technologies and document management systems
Strong attention to detail and organizational skills are essential for success in this role.
Work Location
The job is based in Aundh, Pune, one of the major IT and corporate hubs in Maharashtra.
Many pharmaceutical companies, CROs, and healthcare technology firms operate in this region, making Pune an excellent location for professionals pursuing careers in clinical research and pharmaceutical services.
How to Apply
Interested candidates can apply by sending their updated resume to the recruiter.
Recruiter Name: Hritik Kale
Email: hritik.kale@transperfect.com
Your resume should highlight:
- Clinical research experience
- Knowledge of TMF or eTMF systems
- Regulatory documentation experience
- Relevant software and technical skills
Why Consider This Role?
Here are some key benefits of this opportunity:
- Work with a global clinical research organization
- Gain experience in clinical documentation and regulatory compliance
- Exposure to international clinical trials
- Opportunities for career growth in the CRO industry
- Access to learning and development programs
For professionals aiming for roles such as:
- Clinical Trial Associate
- TMF Specialist
- Regulatory Affairs Specialist
- Clinical Operations Manager
This position can serve as an excellent career stepping stone.
Career Scope in Clinical Documentation
The demand for clinical documentation specialists has grown significantly due to the expansion of global clinical trials and digital trial management systems.
With experience, professionals can advance to roles such as:
- Senior Clinical Document Specialist
- TMF Manager
- Clinical Trial Associate
- Clinical Operations Manager
- Regulatory Affairs Specialist
The clinical research industry continues to expand worldwide, offering stable and well-paying career opportunities.
Final Thoughts
The Clinical Document Specialist I role at TransPerfect is a great opportunity for professionals with clinical research experience who want to specialize in clinical documentation and trial master file management.
If you have strong attention to detail, regulatory knowledge, and experience handling clinical trial documents, this job could be the perfect next step in your career.
Apply soon and ensure your resume highlights your clinical documentation and regulatory expertise.