The clinical research industry in India continues to grow rapidly, opening doors for talented professionals who want to build a career in clinical trials and drug development. If you are an early-career clinical research professional, here’s an exciting opportunity for you.
AnaZeal Analyticals and Research Pvt. Ltd. is currently hiring a Clinical Research Associate (CRA) in Navi Mumbai. This role is ideal for candidates with 0.6 to 2 years of experience in clinical research or site monitoring who are eager to advance their careers in a reputed Clinical Research Organization (CRO).
Let’s explore the complete details of this opportunity.
About AnaZeal Analyticals and Research Pvt. Ltd.
AnaZeal Analyticals is a recognized name in the Bioavailability/Bioequivalence (BA/BE) and clinical research sector in India. The organization offers comprehensive services related to clinical trials, bioanalytical studies, and pharmacovigilance.
Working with AnaZeal gives professionals exposure to:
- Real-world clinical trial monitoring
- Advanced clinical research methodologies
- Strong regulatory compliance practices
- Collaboration with experienced investigators and research teams
For aspiring clinical research professionals, this company provides a strong platform to gain hands-on experience in the CRO industry.
Job Overview
Position: Clinical Research Associate (CRA)
Company: AnaZeal Analyticals and Research Pvt. Ltd.
Location: Navi Mumbai, Maharashtra
Experience Required: 0.6 – 2 Years
Industry: Clinical Research / CRO
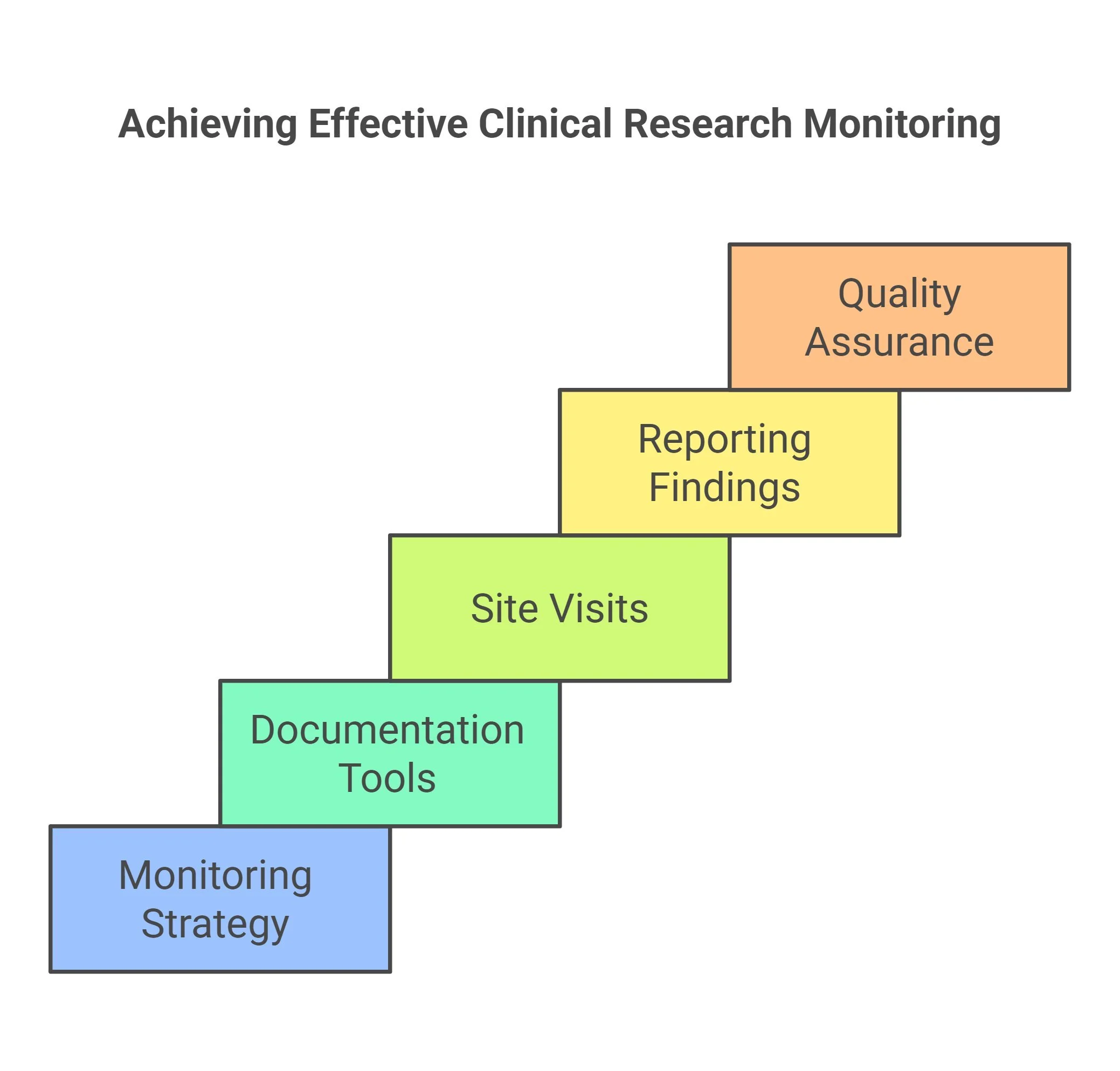
Key Responsibilities of the CRA Role
As a Clinical Research Associate, you will play a critical role in ensuring that clinical trials are conducted ethically, safely, and according to regulatory standards.
Your responsibilities will include:
Site Monitoring and Management
- Conduct regular site monitoring visits
- Ensure trials follow the approved protocol and regulatory guidelines
Source Data Verification (SDV)
- Verify clinical trial data by comparing Case Report Forms (CRFs) with original medical records
- Maintain data accuracy and integrity
Regulatory Compliance
- Ensure all trial activities comply with ICH-GCP guidelines
- Maintain proper documentation and regulatory records
Investigator Coordination
- Act as the primary communication link between investigators, sponsors, and study teams
- Resolve study-related queries and provide guidance
Travel for Site Visits
- Travel to clinical sites for monitoring visits and audits as required
Eligibility Criteria
AnaZeal is looking for motivated early-career professionals with a strong understanding of clinical research processes.
Educational Qualification
Candidates with the following backgrounds can apply:
- Life Sciences
- Pharmacy
- Clinical Research
Experience
- 0.6 to 2 years of experience in clinical research or CRA role
Technical Skills
- Strong understanding of ICH-GCP guidelines
- Knowledge of clinical trial monitoring and documentation
Soft Skills
- Excellent communication skills
- Strong organizational and reporting abilities
- Ability to manage multiple sites effectively
Gender Preference: Male candidates (as mentioned in the official requirement)
Why Work in Navi Mumbai?
Navi Mumbai is rapidly becoming a major pharmaceutical and clinical research hub in India. The region hosts several CROs, pharma companies, and research laboratories.
Benefits of working here include:
- Close proximity to major pharmaceutical headquarters
- Growing opportunities in clinical research and drug development
- Excellent urban infrastructure and connectivity
For clinical research professionals, Navi Mumbai offers strong career growth potential.
How to Apply
If you meet the eligibility criteria and want to advance your career in clinical research monitoring, you can apply directly.
Application Email:
📧 hr@anazeal.com
Subject Line Suggestion:
Application for CRA – Your Name
Application Tips
- Update your CV with clinical monitoring experience
- Highlight your knowledge of ICH-GCP guidelines
- Mention site monitoring visits or clinical trial experience